OK people, line up to spank me. I was 180 degrees off on this one. The broad-spectrum MMP-inhibiting drugs cause tendinitis by taking out sheddases in addition to MMPs. MMPs appear to be bad for tendons.
Matrix Metalloproteinases: Pro- and Anti-Angiogenic ActivitiesControlling for systemic dose and inhibitor potency, the MMP-selective (collagense-, gelatinase-) inhibitors caused less tendinitis but were also less effective as anticancer agents. Interestingly, at equivalent dosages, one of the broad spectrum MMPI tested that had less ability to inhibit certain MMP-like enzymes known as membrane protein ''sheddases'' also exhibited less capability to induce tendinitis. ''Sheddases'' are metalloenzymes that release membrane-associated proteins, some of which are growth factors and cytokines such as transforming growth factor (TGF-alpha) (Arribas et al. 1996) and tumor necrosis factor (TNF-alpha), and have previously been shown to be inhibited by certain MMPI (Gearing et al. 1994).
J Cell Biochem. 2005 Sep 1;96(1):117-25.
Mechanical loading stimulates ecto-ATPase activity in human tendon cells.
Tsuzaki M, Bynum D, Almekinders L, Faber J, Banes AJ.
Department of Orthopaedics, University of North Carolina at Chapel Hill, Chapel Hill, North Carolina 27599-7055, USA. Mari_tsuzaki@med.unc.edu
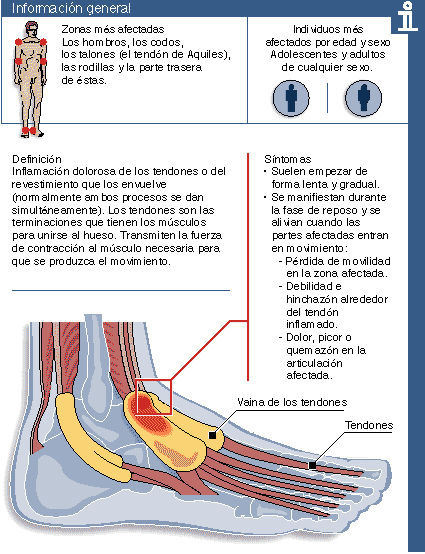
Response to external stimuli such as mechanical signals is critical for normal function of cells, especially when subjected to repetitive motion. Tenocytes receive mechanical stimuli from the load-bearing matrix as tension, compression, and shear stress during tendon gliding. Overloading a tendon by high strain, shear, or repetitive motion can cause matrix damage.
Injury may induce cytokine expression, matrix metalloproteinase (MMP) expression and activation resulting in loss of biomechanical properties. These changes may result in tendinosis or tendinopathy. Alternatively, an immediate effector molecule may exist that acts in a signal-dampening pathway. Adenosine 5'-triphosphate (ATP) is a candidate signal blocker of mechanical stimuli. ATP suppresses load-inducible inflammatory genes in human tendon cells in vitro. ATP and other extracellular nucleotide signaling are regulated efficiently by two distinct mechanisms: purinoceptors via specific receptor-ligand binding and ecto-nucleotidases via the hydrolysis of specific nucleotide substrates. ATP is released from tendon cells by mechanical loading or by uridine 5'-triphosphate (UTP) stimulation. We hypothesized that mechanical loading might stimulate ecto-ATPase activity. Human tendon cells of surface epitenon (TSC) and internal compartment (TIF) were cyclically stretched (1 Hz, 0.035 strain, 2 h) with or without ATP. Aliquots of the supernatant fluids were collected at various time points, and ATP concentration (ATP) was determined by a luciferin-luciferase bioluminescence assay. Total RNA was isolated from TSC and TIF (three patients) and mRNA expression for ecto-nucleotidase was analyzed by RT-PCR. Human tendon cells secreted ATP in vitro (0.5-1 nM). Exogenous ATP was hydrolyzed within minutes. Mechanical load stimulated ATPase activity. ATP was hydrolyzed in mechanically loaded cultures at a significantly greater rate compared to no load controls. Tenocytes (TSC and TIF) expressed ecto-nucleotidase mRNA (ENTPD3 and ENPP1, ENPP2). These data suggest that motion may release ATP from tendon cells in vivo, where ecto-ATPase may also be activated to hydrolyze ATP quickly. Ecto-ATPase may act as a co-modulator in ATP load-signal modulation by regulating the half-life of extracellular purine nucleotides. The extracellular ATP/ATPase system may be important for tendon homeostasis by protecting tendon cells from responding to excessive load signals and activating injurious pathways.
PMID: 16052485